
News
- Home
- News


Massivit3D to go public on Tel Aviv Stock Exchange via IPO at $200 million valuation
Israeli large-format 3D printing firm Massivit3D is set to go public on the Tel Aviv Stock Exchange (TASE) imminently, sources “close to the matter” have told Globes.
Before the Initial Public Offering (IPO) goes ahead, market sources believe that Massivit3D aims to raise $50 million in funding, in a deal valuing the company at $200 million. Although it’s currently unclear where this investment will come from, interior design firm Klil Industries and 3D printer manufacturer Stratasys have provided funding in the past.
“In future, one of the main barriers to adopting 3D printing will be speed,” said Erez Zimerman, CEO of Massivit3D. “That’s why Massivit has developed our technology, in which we can print at least 30 times faster than related systems, and this will allow more and more companies to adopt 3D printing on the industrial side.”

Massivit’s 3D printing technology
Founded in 2013 and based in the Israeli city of Lod, Massivit3D manufactures and markets large-format 3D printers, in addition to an accompanying array of materials and software. The company’s product range is essentially designed to enable clients in the engineering and architectural markets to create scale one models and parts quickly and cost-effectively.
Massivit3D’s newest system, the ‘Massivit3D 10000,’ is set to be launched in fall 2021, and is built to address the tooling requirements of those in the automotive and aerospace sectors. The printer operates using the firm’s Cast-In-Motion (CIM) technology, in which a gel is cured, cast into shape, and immersed in water to allow sacrificial materials to break off.
The machine features a large build volume, that enables users to fabricate complex tooling designs in vast numbers, while accelerating the process of iterating new product designs. Although Massivit3D hasn’t specified the exact build area of its upcoming machine, it does claim that it’s “at least 30 times faster” that similar casting-based systems.
At present, the company holds 52 technology patents, and it has received five contingent purchase orders from clients in the U.S, U.K, France and Taiwan. With its application to be listed on the TASE, Massivit3D is now attempting to accelerate the commercialization of its technology, and expand further into the architecture and renewable energy markets.
The $200 million TASE IPO
Given that Massivit3D isn’t a publicly-listed company just yet, there’s very little information about its finances in the public domain. However, it’s understood that the firm has raised around $20-$30 million in previous funding rounds, with investors including Klil’s Zvi Neta and Tzuri Daboosh as well as Stratasys.
Whether Massivit3D’s IPO is financed by these investors again or the company finds new backers remains to be seen, but it has been confirmed that equity firm Poalim IBI will underwrite the offering. Massivit3D’s valuation appears to be based on its earnings and technological potential, and it reportedly turned over $50 million in revenue from 2017 to 2019.
Although it has also been reported that the company’s sales in 2020 were impacted by the pandemic, it’s now understood to be entering revenue recovery like the rest of the 3D printing industry. 3D Systems, for instance, recently posted strong initial Q4 2020 results, while PyroGenesis’ financial guidance projected growth of over 300%.
Massivit3D was also co-founded by Gershon Miller, a seasoned entrepreneur who has sold firms like Idanit to Scitex before, and most recently sold Objet to Stratasys. As a result, the company’s offering appears to be in safe hands, while more generally, its progress reflects a growing industry enthusiasm for IPOs.
Desktop Metal went public on the NYSE last year after a merger with Trine Acquisition, which saw the company raise $580 million in capital, effectively funding its $300 million acquisition of EnvisionTEC. Israeli tech firms have also been active on the TASE recently, with additive food company Meat-Tech 3D filing for an IPO in November 2019.
3D printing on a grand scale
Massivit3D isn’t the only company that manufactures large-format 3D printers, and a range of other scalable systems have developed in recent years.
Intech Additive Solutions, for instance, has launched its large-format iFusion LF series of metal 3D printers. The machines’ 450 x 450 x 450mm build volumes and broad metal compatibility are geared towards Indian manufacturers operating in the aerospace and automotive sectors.
Last year at Formnext Connect, German 3D printer manufacturer SLM Solutions launched its large-format challenger with its new NXG XII 600 system. The machine includes a huge 600 x 600 x 600 mm build envelope and twelve optimized 1 KW lasers, that allow it to address the needs of large-volume serial production clients.
Elsewhere, in a more application-focused approach, 3D Systems is developing the “world’s largest 3D printer” for the U.S. Army. The machine’s 1m x 1m x 600mm build area is designed to enable the fabrication of parts that address the ammunition, vehicle, helicopter, and missile defense demands of the Armed Forces.
Please do read the original article by Paul Hanaphy here.

PostProcess Technologies Live Solution Experience Tours in 2021
Join PostProcess Technologies for a Live Solution Experience tour!
Sign up today for a PostProcess Live Solution Experience, a real-time group tour of automated 3D post-printing focused on key print technology applications. Conducted by an expert Engineer broadcasting from our lab, you will get a close-up view of how full-stack technology encompassing software, hardware, and chemistry works. See the solutions running live on the proprietary AUTOMAT3D™ platform and engage in real-time Q&A. Explore options for various sessions below!
Are you interested to learn more about PostProcess Technologies and the results they provide?
Please follow the link for FDM & Polyjet here or for Resin Removal here.

Desktop Metal Expands Its Production System Lineup with New Printer Designed to Bridge Process Development and Full-scale Metal Parts Mass Production
The New P-1 Joins the Production System P-50 to Offer Single Pass Jetting Technology for Process Development and Serial Production Applications
- Expanding the Production SystemTM lineup, the new P-1 bridges benchtop process development and mass production for customers looking to scale to an industrial, high-volume additive manufacturing capacity
The P-1 offers Desktop Metal’s patent-pending Single Pass JettingTM (SPJ) technology in an inert environment with a smaller 1-liter form factor designed to print a full layer in less than 3 seconds
Full P-1 builds can be printed in less than one hour and process parameters transfer directly to the P-50, making the P-1 an ideal tool to develop materials and validate new applications without taking P-50 hardware offline from mass production jobs; the P-1 is also well-suited for running serial production of small, complex parts
Desktop Metal has begun shipments of the P-1, with Ford Motor Company as a key initial customer
The P-50, the flagship Production System printer, is designed to achieve speeds up to 100 times those of legacy powder bed fusion (PBF) additive manufacturing technologies(1), remains on schedule to begin volume commercial shipments in 2021, paving the way for the mass production of end-use parts, and unlocking throughput, repeatability, and part costs competitive with conventional mass production techniques
December 17, 2020, BOSTON, MA – Desktop Metal (NYSE: DM), a leader in mass production and turnkey additive manufacturing solutions, today announced the new P-1 printer has begun global shipments and joins the Production SystemTM lineup alongside the flagship P-50 printer. Designed to serve as a bridge from process development to full-scale mass production of end-use metal parts, the P-1 leverages the same patent-pending Single Pass JettingTM (SPJ) technology and core additive manufacturing benefits for companies and research institutions alike at the size and scale of serial production. The P-1 is now available for order and has already begun to ship to initial customers, including to the Ford Motor Company, which will receive its printer this month.

“We know industrial businesses around the world are eager to begin working with the Production System P-50 and benefit from the fastest, most cost-effective way to manufacture metal parts of all levels of complexity at-scale,” said Ric Fulop, CEO and co-founder of Desktop Metal. “Adding the P-1 to our Production System portfolio serves as a key enabler for these companies as they look to develop processes and materials on a smaller scale before ramping up to mass production volumes. Similarly, many businesses and research institutions are also interested in leveraging the economics and quality of SPJ technology for mid-volume serial production, making the P-1 an ideal fit and a great stepping-stone to broad adoption of Desktop Metal’s technology and flagship P-50 printer.”
The Ford Motor Company is among the first early adopters to purchase the new Production System P-1.
“Ford has been active in 3D printing since 1988 with acquisition of the third commercially available stereolithography (SLA) system; we are very excited to be early adopters of the P-1,” said Cynthia Flanigan, Director, Vehicle Research and Technology, Ford Research and Advanced Engineering. “We expect that this new system will serve as an important tool in the development of our future advanced process and alloy implementation, enabling our researchers to investigate additional production opportunities of metal binder jetting at Ford Motor Company. Our early collaboration with Desktop Metal highlighted the need for a lab scale system that is aligned with the functionality of the production scale system so we can further develop expertise around this process.”
A Shared SPJ Technology Architecture Enables Direct Process Transfers Between the P-1 and P-50
Created by leading inventors of binder jetting and single-pass inkjet technology, the Production System P-50 is an industrial manufacturing solution designed to achieve speeds up to 100 times those of legacy PBF additive manufacturing technologies(1), enabling production quantities of up to millions of parts per year at costs competitive with conventional mass production techniques.
The P-1 offers a new form factor to bridge the gap between benchtop process development and mass production, leveraging the same patent-pending SPJ technology and print carriage design as on the P-50 but with enhanced process flexibility. Also similar to the P-50, the P-1 features a state-of-the-art print bar with native 1200 dpi, advanced printhead technology that supports a wide variety of binders, and an inert processing environment to support both non-reactive and reactive materials, a key benefit for businesses and research institutions looking to experiment with a variety of materials. As a result, materials research and new application development conducted on the P-1 can be transferred directly onto the P-50 to scale to mass production, without the need to take this more industrial manufacturing solution offline for R&D activities, enabling efficient process development and new product introductions.
“For many businesses like Ford, the P-1 will serve as a learning lab for processes as they look to scale up to full production,” said Fulop. “Research institutions are also eager to adopt the Production System P-1 to experiment, validate materials, and test a variety of use cases for metal binder jetting with a smaller build box that offers all of the benefits of SPJ technology, including speed, quality, and reliability.
The P-1 Offers Cost-Effective, Serial Metal Parts Production for Small & Complex Parts
SPJ technology on the P-1 is designed to print each layer in less than three seconds, including powder deposition, powder compaction, anti-ballistics, binder deposition, and printhead cleaning. At this maximum build rate, the P-1 can achieve production throughputs 10 times higher than those of legacy PBF systems(1) and fast enough to complete a full build in less than one hour. The P-1’s open material platform and inert process environment allow customers to use low-cost, third-party metal injection molding powders across a variety of materials, making the P-1 suitable for cost-effective serial production of small and complex parts in addition to smaller scale process development activities. Powder reclaimed during the printing and depowdering process can be recycled for future use, driving further cost efficiencies and resulting in a more environmentally friendly manufacturing process. In addition, the tooling-free manufacturing process on the P-1 facilitates quick turnovers to new jobs along with the ability to print many complex geometries simultaneously with no print supports required.

P-1 customers will also gain access to Desktop Metal’s Fabricate® manufacturing build preparation software, as well as to the Company’s newly-released Live Sinter™ application, which dynamically simulates the sintering process and automatically generates print-ready geometries that compensate for the shrinkage and distortion that take place during sintering, minimizing process trial and error while improving accuracy.
The flagship Production System printer, the P-50, remains on schedule to begin volume commercial shipments in 2021, paving the way for the mass production of end-use parts, and unlocking throughput, repeatability, and competitive part costs. For more information on the P-1, the P-50, and Production System technology, visit https://www.desktopmetal.com/products/production.
Please do read the original Press Release by Desktop Metal here.

Leveraging DfAM for Enhanced FDM Post-Printing Efficiencies with PostProcess Technologies
CONTENTS
I. Introduction to Design for Additive Manufacturing
II. FDM Background
III. DfAM Strategies
IV. Results
V. Conclusions
I. INTRODUCTION TO DESIGN FOR ADDITIVE MANUFACTURING
Additive manufacturing (AM) is a powerful technology that lends itself to producing organic geometries, and building parts in short timeframes. Additive technologies are capable of printing shapes that cannot be created by any other means. One of the most salient factors in successfully building these unique designs is the utilization of soluble support material.
As is well-known by anyone who has worked with additive technology, 3D-printed end use parts and prototypes do not come off of the printer “customer-ready.” Virtually every printed part, regardless of print technology, requires some sort of post-printing, whether that be support removal, resin removal, surface finish, or all the above.
As the additive production process has been classically categorized into three separate silos; design, build/print, and post-print, the final step is all too often an afterthought. Rather than considering the additive process a linear, sequential one, PostProcess Technologies embraces the ideology that efficiencies and end part results can be dramatically improved when all of the steps are conceptualized as interdependent.This integrated approach is an advanced ideology that will be necessary to scale additive’s impact, and eventually usher in Industry 4.0.
This paper will identify the ways in which designing for additive manufacturing (DfAM) can affect efficiencies, cost, and support material usage in Fused Deposition Modeling (FDM) printing. Print orientation, support settings within the slicing software, and part design are all aspects which can be leveraged to streamline additive workflows, and facilitate faster, more cost-effective post-printing.

It should be noted that while some of these DfAM for FDM techniques can be applied to other print technologies, this is not the case universally (e.g. self-supporting angles are also useful in DfAM for stereolithography (SLA), but not selective laser sintering (SLS) or PolyJet).
As a rule of thumb, DfAM will have a greater effect in circumstances where there is a higher level of design freedom. While DfAM will have a minimal impact on rapid prototyping or tooling applications (situations with the lowest allowance for design freedom) manufacturing aides and end-use production parts allow for more unique designs, thus making DfAM more significant. The more design principles that are considered upfront, the less time must be later allotted to redesign, or the removal of unnecessary support structures later in the process.
II. FDM BACKGROUND
To comprehend the DfAM techniques that will streamline an FDM workflow, it is vital to comprehend the advantages and limitations of the print technology, and why it is one of the most popular 3D printing technologies available today. The process starts with a thermoplastic filament wound in spools. This filament could be ABS, Polycarbonate, or Ultem™*, for example (see the image below for a complete list of Stratasys thermoplastic materials available for use with the brand’s FDM printers). Strength requirements, the temperature that the part will be exposed to, its performance expectations, and the sort of chemical resistance that it requires are all factors that may impact material selection.

The filament is extruded through a heater block, then a nozzle, and computer-controlled motion is used to deposit the melted thermoplastic. As the deposited plastic cools, the z-stage build platform is lowered to create the 3D part. While this layer-by-layer deposition process is ideal for parts with low heights, FDM printers can face challenges printing complex geometries with tall and thin walls. Virtually anywhere there is an overhang on an FDM part, support material will likely be required to prevent build failure, leading to high support material usage. Consequently, FDM processes involve tedious support removal steps, which traditionally involve dissolving support material by using detergents or breaking the supports away manually, depending on the support material used.
As reported by the PostProcess Technologies’ 2020 Annual Additive Post-Printing Trends Report, material extrusion printing methods (e.g. FFF, FDM, MEM) are utilized by 71% of additive manufacturing users, making them the most popular print technology. The top challenge of material extrusion technology was found to be the length of time to finish parts, while the runner-up answer was damaged parts, both issues that DfAM can help mitigate. That being said, additional research has found that 23% of the average part cost for polymer 3D printing is attributed to the post-printing step alone (Source: 2019 Wohler’s Report). Finally, keeping support material to a minimum is ideal in lowering the build time required for a part, the material cost of a part, as well as the time and resources which must be allocated to the post-printing step.
III. DFAM STRATEGIES
Orientation
Depending on the geometry of your part, orientation may play a paramount role in DfAM. Usually a sole aspect like part strength, support structures, print speed, or surface quality must be prioritized to determine the best orientation of the part. Strength is most often the driving factor for orientations, though priorities may change depending on print materials used, or time restraints. Considerations regarding part orientation should be explored during the design phase to proactively ensure minimal support material requirements, as well as printing and post-printing speeds.
Figure 1 shows a L-bracket printed on its end and Figure 2 shows the same L-bracket printed flat on its side. Utilizing GrabCAD Print software, the green represents the model material of the part and the orange represents the support material required to print the part in that orientation. This change in build orientation accounted for a 58% reduction in build time, and a 91% reduction in support material usage, equating to cost, material, and time savings.


Part Design
Knowing the advantages (and limitations) of a print technology is vital in designing the most efficient, structurally-sound part possible.
Self-Supporting Angles
FDM is one of the only technologies that leverages self-supporting angles. Typically, as long as there is at least a 45° overhang to the build platform, support materials will not be needed. This strategy can also come into play by implementing chamfers and/or fillets, for example see Figures 3 and 4. Also by using the SMART support settings within GrabCAD Print or Insight on Stratasys printers, the software will automatically utilize self-supporting angles when developing support structures, which will help to reduce the amount of support material needed in comparison to other support styles. The actual self supporting angle value will vary based on the printed model material and the slice height that the printer is set at. For example, on a Stratasys Fortus 450mc, loaded with ASA material and printing at a slice height of 0.010” / 0.254 mm will have a self-supporting angle of 43°, whereas Nylon 12CF is 50°.


Implementing Strategic Holes
Designing vertical, non-critical holes as diamond or teardrop-shaped instead of round will reduce the amount of support material required (see bearing block image below). Critical diameter holes should be printed in the XY plane if possible. If the vertical holes require tighter accuracy, they may be drilled out in a post-printing step. When using an automated support removal system it is best practice to reduce the amount of blind holes and cavities. Rather, when possible implement through holes and open cavities, allowing the support removal detergent to flow through those areas more freely, and remove support material faster.

Material Usage
In certain circumstances, utilizing model material as support material can decrease build time, specifically by reducing wait time caused by the printer switching between model and support material. However, this time reduction is only realized if the entire layer’s support material can be changed to model. Whenever you slice a part in GrabCAD Print or generate support material in Insight, the software automatically generates support material and will interface directly with the model material, giving it a good foundation to build model material on. When changing the support material to model material, you will want to leave the three or four support material interface layers, or the layers below where the model material starts, as support material. Otherwise, the model support may end up fused to the finished part,
subsequently causing damage to your overall part.
Surface Finishing
Orientation can also impact the surface finish of a part. Exterior contour toolpaths will result in a better surface finish than exposed raster toolpaths, as rasters are more likely to cause air gaps, which can prove difficult to remove. While flat orientations may print faster, particularly with FDM, horizontal orientations with exterior contour paths will take significantly less time to post-print (see example in image below). FDM parts will also always have a “seam”, which is the start/stop point of each build layer. Utilizing the slicing software to determine where a seam should be placed can help reduce surface finishing time on specific part areas.

IV. RESULTS
As a result of implementing the various DfAM techniques that this white paper covers, small-scale FDM parts were able to achieve significant build time and material savings, thus streamlining the entire additive process. Below is a step-by-step explanation of what part features were changed in the CAD modeling software, and what settings were changed in GrabCAD Print to achieve the desired results, reducing build time and support material usage.
Sensor Bracket Part

Part features added:
– Interior and exterior fillets added to increase the strength of the bracket.
GrabCAD Print Settings:
– Orientate the part with the “L” profile in the XY plane of the build platform (see picture below).
– Reduce the amount of support material required by selecting “Do not grow supports” in the Support Settings.
Bearing Block Part

Part features added:
– Modified the mounting holes from round to diamond shaped, making the geometry self-supporting.
GrabCAD Print Settings:
-Orientate the part with the diameter of the large vertical hole in the XY plane of the build platform (see picture below).
– To place the layer seams on the back of the part (a noncritical surface) select the surfaces where you don’t want the seam placed (critical surfaces), and select “Avoid Seams” in the Model Setting section. This will help to reduce surface finishing time on critical surfaces. This feature only works for parasolid part files that are imported into GrabCAD Print, and will not work with STL files.
Coupling Part
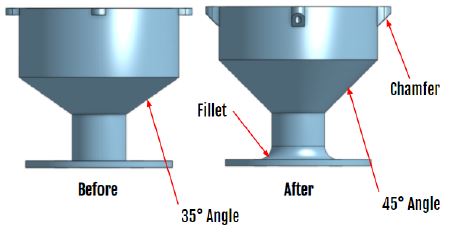
Part features added:
– Changed the angle of the coupling from 35° to 45° to make the geometry self-supporting.
– Added a fillet between the base of the part and the vertical cylinder to help strengthen that area.
– Added a chamber under the top tabs to make the geometry self-supporting.
GrabCAD Print Settings:
– Orientate the part with the round base in the XY plane, as shown below.
Before and After Printing Results
The below picture illustrates the reduction in build time and support material used by making the small design changes and printer setting changes listed above.

Sensor Bracket

Bearing Block

Coupling
Before
Build Time: 2:51
Model: 3.008
Support: 0.658
After
Build Time: 2:25
Model: 3.107
Support: 0.321
Before
Build Time: 5:05
Model: 7.544
Support: 0.742
After
Build Time: 4:18
Model: 7.709
Support: 0.219
Before
Build Time: 4:16
Model: 3.694
Support: 2.092
After
Build Time: 2:47
Model: 3.833
Support: 0.312
Difference
Build Time: -26 minutes
Model: +.01 in³ / +0.25 mm³
Support: -0.34 in³ / -8.64 mm³
Difference
Build Time: -47 minutes
Model: +0.17 in³ / +4.32 mm³
Support: -0.52 in³ / 13.21 mm³
Difference
Build Time: -89 minutes
Model: +0.14 in³ / +3.56 mm³
Support: -1.78 in³ / 45.21 mm³
V. Conclusions
- To utilize additive to the best of its ability, it is essential to recognize that the design, build, and post-print steps are heavily integrated. It is most strategic to consider support removal and surface finishing during the design phase.
- When designing an FDM part for optimized post-printing, factoring in material selection, part orientation, self-supporting angles, contour toolpaths, as well as chamfers and fillets as needed, can contribute to time, cost, and materials savings.
- Automated post-printing solutions from PostProcess Technologies can further enable efficiencies within additive workflows, and cut down on overall time and money spent on the AM process. The proprietary Volumetric Velocity Dispersion technology for soluble support and Suspended Rotational Force for surface finishing both leverage various chemical and mechanical energy sources for optimal post-printing for FDM.
POSTPROCESS TECHNOLOGIES INC.
2495 Main Street, Suite 615
Buffalo NY 14214, USA
+1.866.430.5354
POSTPROCESS TECHNOLOGIES INTERNATIONAL
Les Aqueducs B3, 535 Route des Lucioles
06560 Sophia Antipolis, France
+33 (0)4 22 32 68 13
Please visit PostProcess Technologies for more info and white papers.

Mimaki Illuminates 3D Printing Market with New Compact Full-Colour UV Inkjet 3D Printer
The new Mimaki 3DUJ-2207 3D Inkjet Printer boasts full-colour high definition production in a sleek, compact design, with over 10 million colours
The machine delivers an affordable, scalable solution to drive accessibility to 3D printing and deliver its cutting-edge technologies to a host of new customers
Mimaki Europe, in conjunction with Mimaki USA, a leading manufacturer of inkjet printers and cutting systems, today announces the launch of its new compact, full-colour 3DUJ-2207 UV Inkjet 3D Printer. Previously the first to bring over 10 million colours to the 3D printing market with its larger-scale industrial counterpart, the 3DUJ-553, Mimaki now combines the same impressive colour range and renowned build quality in a compact, affordable solution. With this latest offering, Mimaki aims to extend the reach and accessibility of its cutting-edge 3D printing technologies to an entirely new segment of customers.
The innovative 3D printing solution represents a huge step forward for detailing and post-processing, with the unique combination of its full-colour capabilities and water-soluble support materials enabling super-fine details to be printed in vibrant colour, and then beautifully preserved without the substantial breakage risks usually associated with manual cleaning, painting and finishing. With additional features such as Mimaki’s trademark clear resin, which can be utilised alone or mixed with colours to achieve varying levels of transparency, the new 3DUJ-2207 3D printer presents a robust, advanced 3D printing solution with an affordable price tag – all within a machine sufficiently compact to fit in an office elevator.
“Here at Mimaki, we do not stop at developing disruptive technologies – we make it our business to look even further beyond this, continually striving to find ways in which we can then accelerate the adoption of these technologies and drive the wider industry forward,” comments Danna Drion, Senior Marketing Manager at Mimaki Europe. “Our new 3DUJ-2207 3D Printer is a prime example of this. We had already raised the bar in 3D printing by delivering the world’s first 3D printer with over 10 million colours – but now, with the introduction of our new 3DUJ-2207 3D Printer, we are bringing these 10 million colours to a host of new customers, which in turn means new applications and an even quicker uptake of 3D printing technologies as a whole.”

Set to be commercially available worldwide from January 2021, the 3DUJ-2207 has been designed with functionality at its core, with the compact design and reduced 203 x 203 x 76mm build space just two of many key features which demonstrate its unique versatility and make it ideally suited for office environments. The 3D printer’s quiet performance and optional deodoriser minimise some of the primary disruptions usually associated with 3D printing technologies, ensuring maximum workability in busy workspaces.
Utilising UV-curing inkjet technology, the expansive high-definition colour expression made possible with the Mimaki 3DUJ-2207 3D Printer is around twice that of powder bed manufacturing methods. This provides new possibilities for prototyping and enables the accurate reproduction of subtle colour differences which are critical for many industrial design applications such as medical and architectural modelling. Additional applications include small-scale models for design offices and educational settings, as well as collectible figures.
Drion concludes, “By combining our technological expertise with a wealth of industry experience and market insight, we have been able to create an innovative, inspired solution that merges functionality, affordability and design in a way that really will be game-changing for a lot of creators. This launch will deliver a world of new possibilities to designers and product developers, for many of whom the idea of high-definition full-colour 3D printing might previously have been out of reach, and that is something we are extremely proud of.”
The Mimaki 3DUJ-2207 3D Printer will be exhibited online at Formnext Connect and as part of Mimaki’s latest virtual event, the Mimaki 3D Experience, from 10th November to 16th December. Follow Mimaki’s social media channels for more information.
Please do read the original press release here or visit MimakiEurope.com to learn more.

Envisiontec Speed and Castability: What Factors Affect Final Casting Results?
Over the past few years, 3D printing has been transforming the jewelry industry. The introduction of 3D printers gives jewelers the capability to produce castable, high detailed parts in a fraction of time. But not all 3D printers are created equal, and casting results may vary widely, leaving some custom jewelers and manufacturers frustrated.
Additive manufacturing cuts production time dramatically when compared to traditional hand-carving methods and lately there has been a race for developing faster 3D printers for jewelers to produce rings in a matter of minutes and not hours. But does build speed really matter in 3D printing? Should you have to sacrifice quality to achieve it?
With a long standing history of 3D printing innovation, EnvisionTEC focuses on real solutions to real application problems. The company introduced its patented Continuous Digital Light Manufacturing (cDLM) technology so that jewelry professionals wouldn’t have to choose between speed and quality.

DLP technology, which was first developed and patented by EnvisionTEC, has been the gold standard for 3D printing jewelry. A standard DLP 3D printer builds parts in layers, one layer at a time. Each layer must be exposed to the light source and the time of exposure is defined by layer thickness and material reactivity. Once a layer has been exposed, and the specified layer is formed, a layer separation procedure is performed to peel the layer from the bottom of the material tray and replenish fresh resin to cure the next layer.
Over the years, EnvisionTEC has perfected a number of patented methods of minimizing the effects of this peeling separation, with highly castable options available to the jewelry industry that are widely used from custom designers to large manufacturers. But there are limits to the amount of wax that can be used with DLP 3D printing.
When using wax-based materials, the exposure times are typically three to four times that of regular plastic acrylic polymers. The higher the material castability (i.e. wax content), the slower the exposure time. A number of other 3D printer manufacturers claim that they can produce rings in minutes using a standard layer peeling process. However, their resins contain much more plastic than wax, resulting in less than optimal casts with significant surface roughness and porosity.
EnvisionTEC’s cDLM technology works by adding an air gap (similar to the air gap below the puck on an air hockey table surface) between the part being built and the bottom of the material tray. By building a floating layer, the separation forces are completely eliminated – so there is no peeling at all. With no separation forces, cDLM 3D printers are able to print resins with up to a 90% wax fill (Easy Cast 2.0), allowing for patterns that are as easy to cast as any traditional hand-carved or injection molded wax pattern.

On top of all of this, the cDLM process is fast, allowing jewelers unprecedented manufacturing turnaround times. Traditionally, 3D printing castable materials would take hours to build. However with the cDLM technology, bridal and fashion rings can be designed, grown, and ready for investment casting the same day, which is a huge advantage for the jewelry market.
If you need a solution for 3D printing premium quality wax patterns for jewelry quickly that can also be easily casted, view EnvisionTEC’s cDLM technology. For more information visit: EnvisionTEC Castables.
IT’S SUPER-FAST — CONTINUOUS DIGITAL LIGHT MANUFACTURING
- Parts built as the platform moves continuously without separation, which leads to a drastic reduction in the building time as well as allowing for highly wax-filled resins to be used.
- A drastic reduction in separation, forces delivery of highly accurate parts, with detailed feature resolution.
- Better castability, as the wax based resin is cured twice as much during the build process, when compared to building using non continuous traditional DLP 3D printers. This delivers a porosity-free casting from EnvisionTEC’s ash free castable materials.
- And more rings printed in Easy Cast 2.0 at 15 µm on the EnvisionTEC Vida UHD cDLM.

3D Printed Jewelry | Precise, Fast & Highly Castable from EnvisionTEC on Vimeo.
EnvisionTEC’s combination of 3D printers and its range of exclusive materials provides Isaac with a real competitive edge. Click here to see a video of Isaac Cohen and others describing why they chose cDLM technology.
Please do read the original article by Team EnvisionTEC here or visit EnvisionTEC.com to learn more.

Syqe Medical reaps benefits of sustained high temperature parts with XJet Additive Manufacturing
Rehovot, Israel – November 4th, 2020 – XJet Ltd. has revealed today how Syqe Medical, the manufacturer of a revolutionary medical inhaler, is using XJet NanoParticle Jetting™ (NPJ) technology to create parts impossible to produce with other manufacturing methods.
Syqe Medical’s Selective-Dose inhaler aims to increase the effectiveness of patient treatment by delivering a range of drugs in breakthrough precision levels. This new level of precision allows patients to reach the coveted optimum balance between symptom relief and adverse events, and regain their quality of life.
Finding a need to build a high-precision, high-temperature and electrically insulated test facility for product development, Syqe Medical first looked to PEEK materials and traditional manufacturing methods because of the heat resistance requirements. However, according to Syqe Medical Founder and CEO Perry Davidson, design processes were constrained, requiring many adjustments and consequently, production costs were high, delivery times long and yet still the materials were not durable enough.


“Realising we needed another solution, we turned to XJet,” elaborates Itay Kurgan, Product manager at Syqe Medical. “We have a wealth of experience with additive manufacturing technologies and whilst the polymer materials don’t have the heat resistance we needed, XJet ceramic materials are resistant to temperatures even higher than our requirements and of course they are electrically insulated. Minor design adjustments are very easy, and results are precise and repeatable, so we can achieve optimum accuracy and delivery times are very fast. Where all other options were flawed in at least one aspect, XJet provided the perfect solution.”
The XJet Carmel 1400 AM system – featuring the company’s patented NanoParticle Jetting technology – enables the production of ceramic parts with the ease and versatility of inkjet printing. Using ultrafine layers, the Carmel system produces ceramic parts with superfine details, smooth surfaces and high accuracy which are preserved with the use of soluble support material which simply washes away.
“Syqe Medical is making life-changing advancements in the field of patient care so we’re delighted to see them reaping the rewards of XJet’s unique capabilities. Ceramics have some very valuable material properties, but it can be difficult to exploit them due to difficulties in the manufacturing process. XJet delivers all the benefits of ceramic materials, with accuracy and precision, but without the difficulties of traditional manufacturing,” says XJet CBO Dror Danai.
Please do read the official press release here.

New Product Launch: DEMI 4000 Resin Removal Solution
PostProcess Technologies has once again revolutionized resin removal for additive manufacturing. Leveraging proven submersion technology, the DEMI 4000™ is the world’s only large scale software-driven 3D post-printing solution for high-volume stereolithography (SLA) production. Read on to learn what sets it apart.
Powered Lift System (Key Benefits – Safety and Ease of Use)
The DEMI 4000’s powered lift system allows for automatic lowering and heightening of tray loads. This ergonomic feature saves floor space and messy bath transfers by eliminating the need for multiple machines, and allows for same-height loading of multiple build trays and consistent, operator-free processing.
Large Processing Tank (Key Benefits – High Volume Production)
Measuring 890mm x 890mm x 635mm, the DEMI 4000’s tank is designed to hold 275 gallons of resin removal chemistry. At this size, the tank can handle heavy loads of parts at one time, effectively increasing throughput and improving cycle times for all part sizes. Additionally, as a fully-enclosed system with a ventilation port, the DEMI 4000 ensures safety for operators and manages the work environment to control fumes and contain resin and detergent.


AUTOMAT3D™ Software Control (Key Benefits – High Volume Production)
AUTOMAT3D™ software takes the guesswork out of post-printing and reduces operator labor time with pre-programmed recipes. The software controls the agitation intensities, temperatures, process times, and more to deliver consistently complete resin removal. AUTOMAT3D optimizes the rate of resin removal with advanced ultrasonics, an advanced pumping scheme, as well as heat and variable fluid flow. The system was designed with optimal operator ergonomics including multi-touch HMI with glass screen and stainless steel wash-down control buttons and interface panel.
Additive Formulated Chemistry (Key Benefits – Longevity, Cycle Times)
PostProcess offers a comprehensive suite of detergents, each uniquely optimized for post-processing of numerous resins from various manufacturers. This extends to specialized applications such as ceramic-filled resins and high-temp resins.
Our newest generation of detergent was found to have better longevity than all typical solvents (e.g. IPA, TPM, DPM), and an exceedingly safe flashpoint over 200°F / 93°C. This equates to less frequent chemical change-outs, improved environmental friendliness, and compliance with regulatory requirements.
Printer Alignment
For reference, the DEMI 4000 aligns with the following large SLA printers:
- 3DS ProX 800
- Max build envelope: 6” x 29.5” x 21.6” (650mm x 750mm x 550mm)
- RPS NEO800
- Max build envelope: 5” x 31.5” x 23.6” (800mm x 800mm x 600mm)
- SSYS V650
- Max build envelope: 20” x 20” x 23” (508mm x 508mm x 584mm)
For more info please contact us or visit: https://www.postprocess.com/

Desktop Metal to become public, creating the only listed Pure-Play Additive Manufacturing 2.0 Company
- Desktop Metal is a leader in mass production and turnkey additive manufacturing solutions, offering the fastest metal 3D printing technology in themarket, up to 100x the speed of legacy technologies(1)
- The additive manufacturing industry is estimated to growfrom $12billionto $146billionthis decadeas it shifts from prototyping to mass production
- Desktop Metal to become publicly listed through a business combination with Trine (NYSE:TRNE)
- Combined company to have an estimated post-transaction equity value ofup to$2.5billionand will remain listed onthe NYSE under the ticker symbol “DM” following expected transaction closein the fourth quarter of 2020
- Transaction to provide up to $575millionin gross proceeds, comprised ofTrine’s $300millionof cash held in trust (assuming no redemptions) and a $275M fullycommitted common stock PIPE at $10.00 per share, including investments from Miller Value Partners, XN, Baron Capital Group, Chamath Palihapitiya, JB Straubel, and HPS Investment Partners
- Leo Hindery, Jr., legendary technology investor and operator, to join Desktop Metal’s board
- All significant Desktop Metal shareholders including, Lux Capital, NEA, Kleiner Perkins, Ford Motor Company, GV (formerly Google Ventures), and Koch Disruptive Technologies will retain their equity holdings through Desktop Metal’s transition into the publiclylisted company
BOSTON, MA (August 26, 2020)–Desktop Metal, Inc. (“Desktop Metal” or the “Company”) a leader in mass production and turnkey additive manufacturing solutions, announced today it will become a publicly listed company in order to accelerate its growth trajectory within the rapidly growing additive manufacturing market and capitalize on the strong secular tailwinds supporting the reshoring of manufacturing and supply chain flexibility. The Company has signed a definitive business combination agreement with Trine Acquisition Corp. (NYSE:TRNE), a special purpose acquisition company led by Leo Hindery, Jr., and HPS Investment Partners, a global credit investment firm with over $60 billion in assets under management.Upon closing of the transaction, the combined operating company will be named Desktop Metal,Inc. and will continue to be listed on the New York Stock Exchange and trade under the ticker symbol “DM.”
The additive manufacturing industry grew at a 20 percent annual compound rate between 2006 and 2016 before accelerating to 25 percent compound annual growth over the last 3years, a rate that is expected to continue over the next decade as the market surges from $12 billion in 2019 to an estimated $146 billion in 2030. This market inflection is being driven by a shift in applications from design prototyping and tooling to mass production of end-use parts, enabled by the emergence of what Desktop Metal refers to as “Additive Manufacturing 2.0,” a wave of next-generation additive manufacturing technologies that unlock throughput, repeatability, and competitive part costs. These solutions feature key innovations across printers, materials, and software and pull additive manufacturing into direct competition with conventional processes used to manufacture $12 trillion in goods annually.
Desktop Metal’s cash on hand after giving effect to the transaction will enable the Company to capitalize on its position at the forefront of Additive Manufacturing 2.0 by accelerating the Company’s rapid growth and product development efforts. The Company will also use the proceeds to support constructive consolidation in the additive manufacturing industry.
Led by an experienced team with deep operational and scientific pedigree, Desktop Metal has distribution in more than 60 countries around the world and broad adoption from leading companies spanning array of industries,including automotive, consumer products, industrial automation, medical devices, and aerospace & defense.
Desktop Metal is ready to rapidly deploy its full suite of additive manufacturing solutions to existing and new customers on a global basis. The Company’s broad product portfolio includes the Studio System™, an office-friendly metal 3D printing system for low volume production, which has been shipping in volume for more than a year, as well as the new Shop System™ for mid-volume manufacturing and its continuous fiber composite printer, Fiber™, both of which are expected to ship in the fourth quarter of 2020. The Company’s Production System™, which has begun shipping to early customers and is expected to ship in volume in the second half of 2021, is designed to be the fastest way to 3D print metal parts at-scale, achieving print speeds up to 100x faster than legacy technologies and delivering thousands of parts per day at costs competitive with traditional manufacturing.
“We are at a major inflection point in the adoption of additive manufacturing, and Desktop Metal is leading the way in this transformation,” said Co-founder, Chairman & Chief Executive Officer of Desktop Metal, Ric Fulop. “Our solutions are designed for both massive throughput and ease of use, enabling organizations of all sizes to make parts faster, more cost effectively, and with higher levels of complexity and sustainability than ever before. We are energized to make our debut as a publicly traded company and begin our partnership with Trine, which will provide the resources to accelerate our go-to-market efforts and enhance our relentless efforts in R&D.”
Leo Hindery, Jr., Chairman & Chief Executive Officer of TRNE added, “After evaluating more than 100 companies, we identified Desktop Metal as the most unique and compelling opportunity, a company that we believe is primed to be the leader in a rapidly growing industry thanks to their substantial technology moat, deep customer relationships across diverse end-markets, and impressive, recurring unit economics. Ric has put together an exceptional team and board of directors with whom we are excited to partner to create the only publicly traded pure-play Additive Manufacturing 2.0 company.”
Transaction Overview
Pursuant to the transaction, TRNE, which currently holds $300 million in cash in trust, will combine with Desktop Metal at an estimated $2.5billion pro forma equity value. Assuming no redemptions by TRNE’s existing public stockholders, Desktop Metal’s existing shareholders will hold approximately 74percent of the issued and outstanding shares of common stock immediately following the closing of the business combination.
Cash proceeds in connection with the transaction will be funded through a combination of TRNE’s cash in trust and a$275millionfully committed common stock PIPE at $10.00 per share, including investments from funds and affiliates of Miller Value Partners, XN, Baron Capital Group, Chamath Palihapitiya, JB Straubel, and HPS Investment Partners.
The boards of directors of both Desktop Metal and TRNE have unanimously approved the proposed transaction. Completion of the proposed transaction is subject to approval of Trine and Desktop Metal stockholders and other closing conditions, including a registration statement being declared effective by the Securities and Exchange Commission, and is expected to be completed in the fourth quarter of 2020.
Additional information about the proposed transaction, including a copy of the merger agreement and investor presentation, will be provided in a Current Report on Form 8-K to be filed by TRNE today with the Securities and Exchange Commission and available at www.sec.gov.
Advisors
Credit Suisse is serving as the exclusive capital markets advisor to Desktop Metal and as sole private placement agent to TRNE. BTIG, LLCis serving as financial and capital markets advisor to TRNE. Latham & Watkins LLP is serving as legal advisor to Desktop Metal, and Paul,Weiss, Rifkind, Wharton & Garrison LLPis serving as legal advisor to TRNE. ICR is serving as investor relations and communications advisor to Desktop Metal.
Investor Conference Call
Desktop Metal and TRNE will host a joint investor conference call to discuss the business and the proposed transaction today, August 26, 2020, at 8:00 AM ET. To listen to the conference call via telephone, dial 1-877-407-4018 or 1-201-689-8471 (international callers/U.S. toll) and enter the conference ID number 13708990. To listen to the webcast, please click here. A replay of the call will be accessible at the webcast link.
For Desktop Metal investor relations website,visit www.desktopmetal.com/investors.
About Desktop Metal
Desktop Metal, Inc., based in Burlington, Massachusetts, is accelerating the transformation of manufacturing with an expansive portfolio of 3D printing solutions, from rapid prototyping to mass production. Founded in 2015 by leaders in advanced manufacturing, metallurgy, and robotics, the company is addressing the unmet challenges of speed, cost, and quality to make Additive Manufacturing an essential tool for engineers and manufacturers around the world. Desktop Metal was selected as one of the world’s 30 most promising Technology Pioneers by the World Economic Forum and named to MIT Technology Review’s list of 50 Smartest Companies.
For more information, visit www.desktopmetal.com.
About Trine Acquisition Corp
Trine Acquisition Corp is a blank check company organized for the purpose of effecting a merger, share exchange, asset acquisition, stock purchase, recapitalization, reorganization, or other similar business combination with one or more businesses or entities.
For more information, visit www.trineacquisitioncorp.com.
Forward Looking Statements
This document contains certain forward-looking statements within the meaning of the federal securities laws with respect to the proposed transaction between Desktop Metal, Inc. (“Desktop”) and Trine Acquisition Corp. (“Trine”), including statements regarding the benefits of the transaction, the anticipated timing of the transaction, the services offered by Desktop and the markets in which it operates, and Desktop’s projected future results. These forward-looking statements generally are identified by the words “believe,” “project,” “expect,” “anticipate,” “estimate,” “intend,” “strategy,” “future,” “opportunity,” “plan,” “may,” “should,” “will,” “would,” “will be,” “will continue,” “will likely result,” and similar expressions. Forward-looking statements are predictions, projections and other statements about future events that are based on current expectations and assumptions and, as a result, are subject to risks and uncertainties. Many factors could cause actual future events to differ materially from the forward-looking statements in this document, including but not limited to: (i) the risk that the transaction may not be completed in a timely manner or at all, which may adversely affect the price of Trine’s securities, (ii) the risk that the transaction may not be completed by Trine’s business combination deadline and the potential failure to obtain an extension of the business combination deadline if sought by Trine, (iii) the failure to satisfy the conditions to the consummation of the transaction, including the adoption of the agreement and plan of merger by the shareholders of Trine and Desktop, the satisfaction of the minimum trust account amount following redemptions by Trine’s public shareholders and the receipt of certain governmental and regulatory approvals, (iv) the lack of a third party valuation in determining whether or not to pursue the proposed transaction, (v) the occurrence of any event, change or other circumstance that could give rise to the termination of the agreement and plan of merger, (vi) the effect of the announcement or pendency of the transaction on Desktop’sbusiness relationships, performance, and business generally, (vii) risks that the proposed transaction disrupts current plans of Desktop and potential difficulties in Desktop employee retention as a result of the proposed transaction, (viii) the outcome of any legal proceedings that may be instituted against Desktop or against Trine related to the agreement and plan of merger or the proposed transaction, (ix) the ability to maintain the listing of Trine’s securities on the New York Stock Exchange, (x) the price of Trine’s securities may be volatile due to a variety of factors, including changes in the competitive and highly regulated industries in which Desktop plans to operate, variations in performance across competitors, changes in laws and regulations affecting Desktop’s business and changes in the combined capital structure, (xi) the ability to implement business plans, forecasts, and other expectations after the completion of the proposed transaction, and identify and realize additional opportunities, and (xii) the risk of downturns in the highly competitive additive manufacturing industry. The foregoing list of factors is not exhaustive. You should carefully consider the foregoing factors and the other risks and uncertainties described in the “Risk Factors” section of Trine’s Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, the registration statement on Form S-4 and proxy statement/consent solicitation statement/prospectus discussed below and other documents filed by Trine from time to time with the U.S. Securities and Exchange Commission (the “SEC”). These filings identify and address other important risks and uncertainties that could cause actual events and results to differ materially from those contained in the forward-looking statements.Forward-looking statements speak only as of the date they are made. Readers are cautioned not to put undue reliance on forward-looking statements, and Desktop and Trine assume no obligation and do not intend to update or revise these forward-looking statements, whether as a result of new information, future events, or otherwise. Neither Desktop nor Trine gives any assurance that either Desktop or Trine will achieve its expectations.
Additional Information and Where to Find It
This document relates to a proposed transaction between Desktop and Trine. This document does not constitute an offer to sell or exchange, or the solicitation of an offer to buy or exchange, any securities, nor shall there be any sale of securities in any jurisdiction in which such offer, sale or exchange would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction. Trine intends to file a registration statement on Form S-4 that will include a proxy statement of Trine, a consent solicitation statement of Desktop and a prospectus of Trine. The proxy statement/consent solicitation statement/prospectus will be sent to all Trine and Desktop stockholders. Trine also will file other documents regarding the proposed transaction with the SEC. Before making any voting decision, investors and security holders of Trine and Desktop are urged to read the registration statement, the proxy statement/consent solicitation statement/prospectus and all other relevant documents filed or that will be filed with the SEC in connection with the proposed transaction as they become available because they will contain important information about the proposed transaction.
Investors and security holders will be able to obtain free copies of the proxy statement/consent solicitation statement/prospectus and all other relevant documents filed or that will be filed with the SEC by Trine through the website maintained by the SEC at www.sec.gov. In addition, the documents filed by Trine may be obtained free of charge from Trine’s website at www.trineacquisitioncorp.com or by written request to Trine at Trine Acquisition Corp., 405 Lexington Avenue, 48th Floor, New York, NY 10174.
Participants in the Solicitation
Trine and Desktop and their respective directors and executive officers may be deemed to be participants in the solicitation of proxies from Trine’s stockholders in connection with the proposed transaction. Additional information regarding the interests of those persons and other persons who may be deemed participants in the proposed transaction may be obtained by reading the proxy statement/consent solicitation statement/prospectus regarding the proposed transaction. You may obtain a free copy of these documents as described in the preceding paragraph.
Press Contacts
For Desktop Metal Investor / Media Relations
Lynda McKinney
press@desktopmetal.com
Investor Relations
Mike Callahan / Tom Cook
DesktopMetalIR@icrinc.com
For Trine Acquisition Corp.
Pierre Henry
phenry@trineacquisitioncorp.com
For HPS Investment Partners
Prosek Partners
Mike Geller / Josh Clarkson
mgeller@prosek.com / jclarkson@prosek.com
# # #
(1)Based on published speeds of binder jetting and laser powder bed fusion systems comparable to the Production System™ available as of August 25, 2020and using comparable materials and processing parameters.
Please do read and download the original Press Release by Desktop Metal here.






